
" Now, as for the claim that each fission results in on average 2.5 neutrons being released, this is true, but those diagrams illustrating a nuclear chain reaction are very misleading. Here is another one, is this correct or not?: A typical bomb will use more than 1.5% of the nuclei for fission. That specific number suggests some specific origin where it might apply, it is impossible to tell what is meant here without context. They must be moderated to produce further fission like in an atomic power plant." They cannot possibly collide with and fission anything. When the first (and only?) nucleus of 4x1026 U-235 nuclei in the above bomb fissions (it is split by free neutrons from somewhere - the Polonium-Beryllium initiators?), it will only heat the surrounding as it is not cooled and the 2.5 free neutrons will just fly away and produce nothing with their 2 MeV energy each. This was not really known in the 1940's when the atomic bomb was said to have been invented.ĩ8.5% of the nuclei do not fission as the free neutrons miss them and fly away. For a high-temperature reactor like the liquid-fluoride reactor, graphite (carbon) is used as the moderator. For most reactors, moderation takes place in the water that also cools the reactor. This slowing-down is done by neutrons bouncing off the nuclei of the atomics in the moderating material. 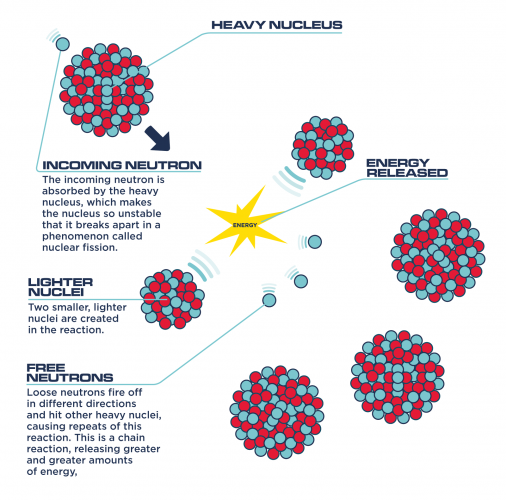 
The moderator in a nuclear reactor is the material whose job it is to slow down neutrons without absorbing them. Actually, for the fissile nuclei such U-235 the SLOWER the neutron is going, the more probable fission is. One might think, intuitively, that if the neutron is going really fast that it has a better chance of “shattering” the nucleus, but that’s not really how it works. It is the speed of the neutron, when it hits the nucleus that has a lot to do with how likely a fission is to occur. The temperature caused by exponential chain reaction will simply melt the uranium-235 (U-235) metal assembly that will flow or evaporate away. 
" So a 52 kg sphere of uranium-235 is a critical mass like 61 kg of rings of uranium-235? What? I just want to know if the person, who said the following, is correct or not? This was not really known in the 1940's when the atomic bomb was said to have been invented." And they have to slow down to have a good chance of causing fission. Actually, the neutrons born from fission are going really fast. And then from fission comes more neutrons, which continue the reaction. So slowed-down neutrons to maximize fission are an absolute requirement. "It is the speed of the neutron, when it hits the nucleus that has a lot to do with how likely a fission is to occur. Of course, there was Oak Ridge, TN, with 75 000 people but they didn't know what they were doing." It is, however 2012 - 67 years later, still TOP SECRET, what US factory managed to separate U-235 from 10 tons of U-238 by gas separation or whatever - magnetism? - and then making it, the U-235, a 72 kg metal slab again and what workshop manufactured and drilled the U-235 metal target rings and projectile ringsin 1945! Reason apparently being that no such workshop or technology existed at that time, 1942-1945, and no rings were ever manufactured. How to separate uranium-235 ( U-235) from uranium-238? To, e.g., obtain 61 kg of pure uranium-235 ( U-235) metal you need about 8 400 kg or 8.4 ton of uranium to separate the uranium-235 ( U-235) from. An uranium-235 ( U-235) atomic has three neutrons less than an uranium-238 atomic. "However uranium-235 ( U235) makes up only 0.72% of normal uranium metal and has to be separated from the remainder (mostly uranium-238) in special factories which makes uranium-235 ( U-235) a little more expensive.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
